
Written by: Senior QA Engineer
Ievgen IevdokymovPosted: 19.03.2026
14 min read
Remote patient monitoring changed care by removing walls. Health is no longer checked occasionally, it’s tracked continuously, in real life.
Heart patches, insulin pumps, and wearables now make clinical decisions outside controlled environments. That’s powerful. And risky.
When software is this close to the patient, failure isn’t acceptable. A brief outage, a data mismatch, or a lost connection can delay action, or miss it entirely. That’s why RPM testing isn’t just about compliance. It’s about trust. And it’s also why these systems are so hard to test.
At DeviQA, we see exactly where RPM solutions fail, and how to prevent it.
Trust in remote patient monitoring begins with QA
Remote patient monitoring shifts healthcare from controlled clinical settings to everyday life. That shift introduces new risks, and removes safety nets.
What makes QA in RPM non-negotiable:
1. Care happens outside hospitals
RPM usage is expanding rapidly: by 2025, an estimated 26 % of the U.S. population (~71 million people) will use some form of RPM services. RPM systems collect continuous physiological data outside clinics, not in predictable, controlled settings.
2. The system becomes the single source of truth
Nearly 50 million Americans already use RPM devices, and clinician adoption has surged, with over 80 % of providers deploying RPM solutions in recent years, meaning clinical decisions increasingly depend on remote data.
3. Small failures have real consequences
RPM interventions have been shown to reduce hospital admissions/readmissions and improve patient safety outcomes in multiple clinical studies, but only when data flow and reliability are preserved.
4. Users are vulnerable by default
RPM adoption is driven by chronic disease management and care for aging populations, key use cases that require continuous monitoring outside clinics to prevent costly readmissions and complications. Population aging trends further increase RPM reliance.
5. Reliability must hold under real-world stress
RPM devices operate over unstable networks and varied environments. Connectivity issues in non-clinical settings are a well-recognized challenge that requires rigorous QA strategies extending beyond basic functional checks.
6. Regulatory compliance is inseparable from quality
Medical device and health software regulators worldwide (e.g., FDA for the U.S., GDPR/HIPAA for data protection) require documented quality, traceability, and risk mitigation as part of compliance, all of which are built on strong QA processes. Regulatory adherence cannot be retrofitted after the fact.
In RPM, quality assurance isn’t a phase or a checkbox. It’s the foundation that determines whether a system can be trusted with patient health.
Engineering trust into remote patient monitoring
Common challenges of RPM software testing
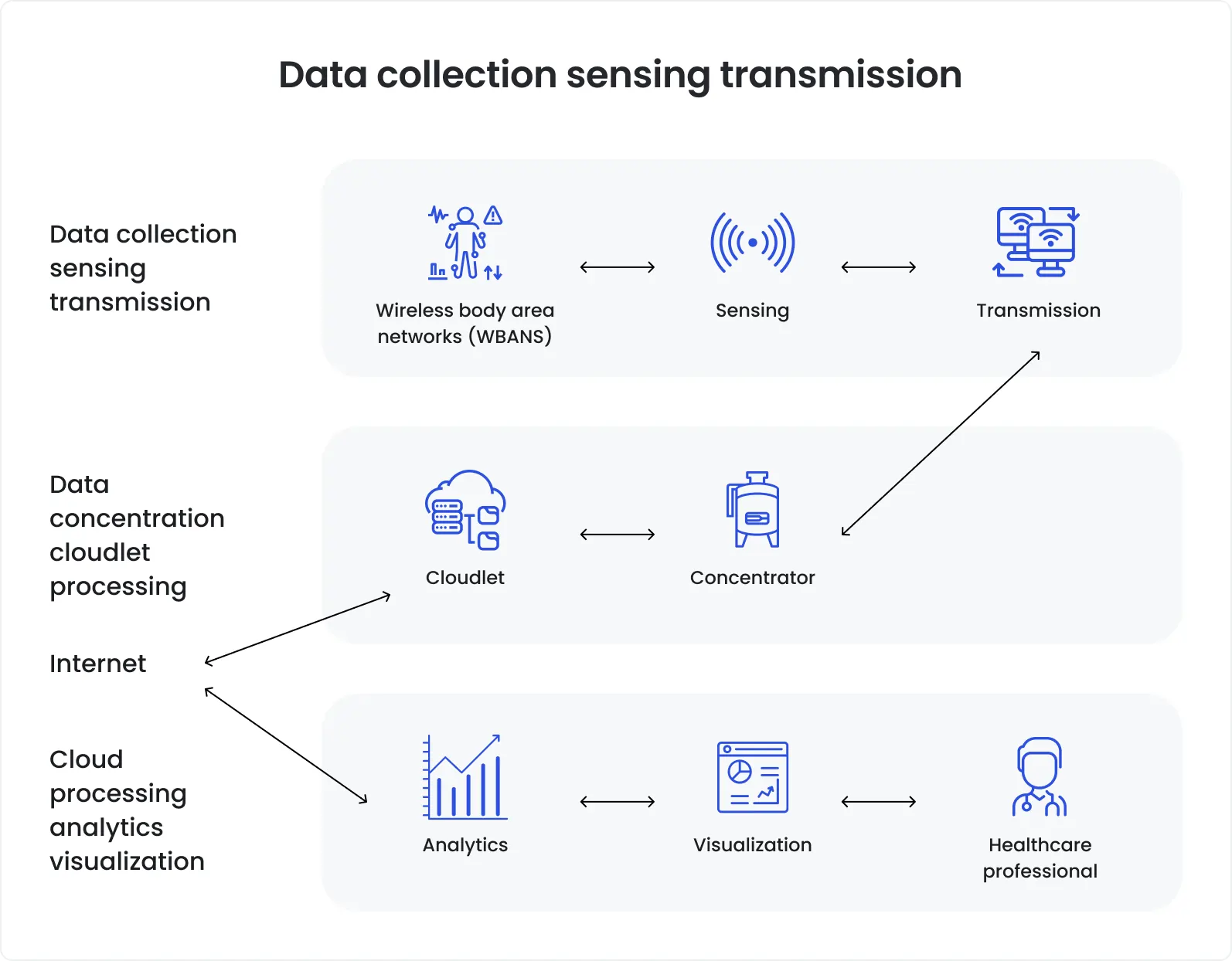
Remote patient monitoring software lives where healthcare is most fragile: in everyday life. It has to work across uncontrolled environments, multiple connected devices, and real-time data flows that clinicians rely on.
That combination makes testing uniquely hard. QA teams aren’t validating a single app, they’re validating an end-to-end system: device → mobile → cloud → (often) EHR, under unstable connectivity and imperfect user behavior, while meeting strict privacy and medical-device expectations.
In practice, most RPM teams run into the same set of QA challenges:
real-world reliability (connectivity, power, wear conditions)
data accuracy and drift over time
security across every layer of the system
regulatory and documentation readiness
accessibility and usability for vulnerable users
regression risk as firmware, apps, and integrations evolve
Let’s break them down.
Challenge 1: Testing RPM devices in real-world environments
Inside hospitals, medical equipment operates in a controlled setting. Power supply is stable, Wi-Fi is managed, and trained medical staff handle devices with care. Remote patient monitoring devices, in turn, are asked to perform in living rooms, cars, gyms, workplaces, or even rural areas, where edge cases are everyday realities. Take unstable Wi-Fi, dropped Bluetooth connections, and inconsistent power sources. Add to this also patients who don’t follow device instructions precisely.
A patient may lose Wi-Fi during a storm or drive through a dead zone, while a Bluetooth connection is meant to be streaming. Sensors may sit loosely on the skin after a workout, or adhesive patches may peel in humid climates. Such a small, seemingly harmless user behavior as putting a phone in battery-saver mode can disrupt the flow of vital data.
All these extraordinary and sometimes unpredictable conditions should be checked in the course of RPM device testing, which is a complex mission.
Tips:
Include in your test matrix environmental variables like weak networks, varying temperatures, sweat, and long wear times in every cycle.
Simulate Wi-Fi drops, mobile handovers, airplane mode mid-stream, and other connectivity disruptions to check recovery and backlog sync.
Test device performance when a device is worn incorrectly, used with a low battery, or misused by patients in other ways.
Ensure the RPM system is resilient enough to indicate low-confidence or corrupted data, preventing distorted readings from silently entering clinical workflows.
Talk through your RPM quality challenges
Challenge 2: Ensuring data accuracy
Data accuracy has always been a top concern in medical software testing. When it comes to RPM systems, this aspect becomes even more critical, as the credibility of such devices heavily depends on the reliability of the data they produce. One inaccurate reading can lead to misdiagnosis or even missed critical events.
A heart-rate monitor that drifts five beats per minute may seem harmless until those errors accumulate across weeks of monitoring and affect a physician’s interpretation of the treatment plan efficiency.
In these conditions, data validation is an essential but daunting task. RPM devices collect biometric and sensor data around the clock, across weeks or months. This stream-based nature introduces several challenges:
1. Data drift: Sensor calibrations often shift after long-term usage, leading to biases that compound over time.
2. Signal degradation: Adhesives loosen, batteries fade, or environmental noise creeps in, quietly lowering accuracy.
3. Historical reliability: For chronic conditions, data have to be always correct so that trends are clinically trustworthy.
Because of this, it’s dangerous to test data accuracy once at device launch. It must be checked throughout development and updates.
Tips:
Check sensor accuracy not only at the point of calibration but also over time, under wear-and-tear conditions.
Implement regression testing for biometric algorithms to detect data drift caused by firmware or software changes.
Automate checks for bias drift, outliers, and degradation for triggering an investigation.
Stress-test long-duration monitoring sessions to spot cumulative accuracy issues that emerge only after long-time device operation.
Test signal integrity when environmental noise, be it motion, sweat, or temperature changes, interferes with readings.
Use controlled baselines to compare historical accuracy, ensuring the alignment of today’s device readings with previous benchmarks.
Make sure that timestamps, units, and identifiers remain intact across device, app, and cloud layers.
Challenge 3: Securing health data across all system layers
RPM solutions collect and store huge amounts of personal data. Let's take Dexcom, a leading provider of glucose monitoring systems. Its privacy policy implies gathering information about an IP address, computer, Internet service, the browser used, and the user’s activities while using Dexcom products and services.
Ensuring that all that personal and health data is secured is one of the most critical remote patient monitoring challenges. The thing is that RPM systems consist of several interconnected layers: the device, the companion mobile app, the cloud, and often the EHR. Each of these layers introduces its own vulnerabilities. If left unchecked, they become weak points that hackers can leverage as loopholes.
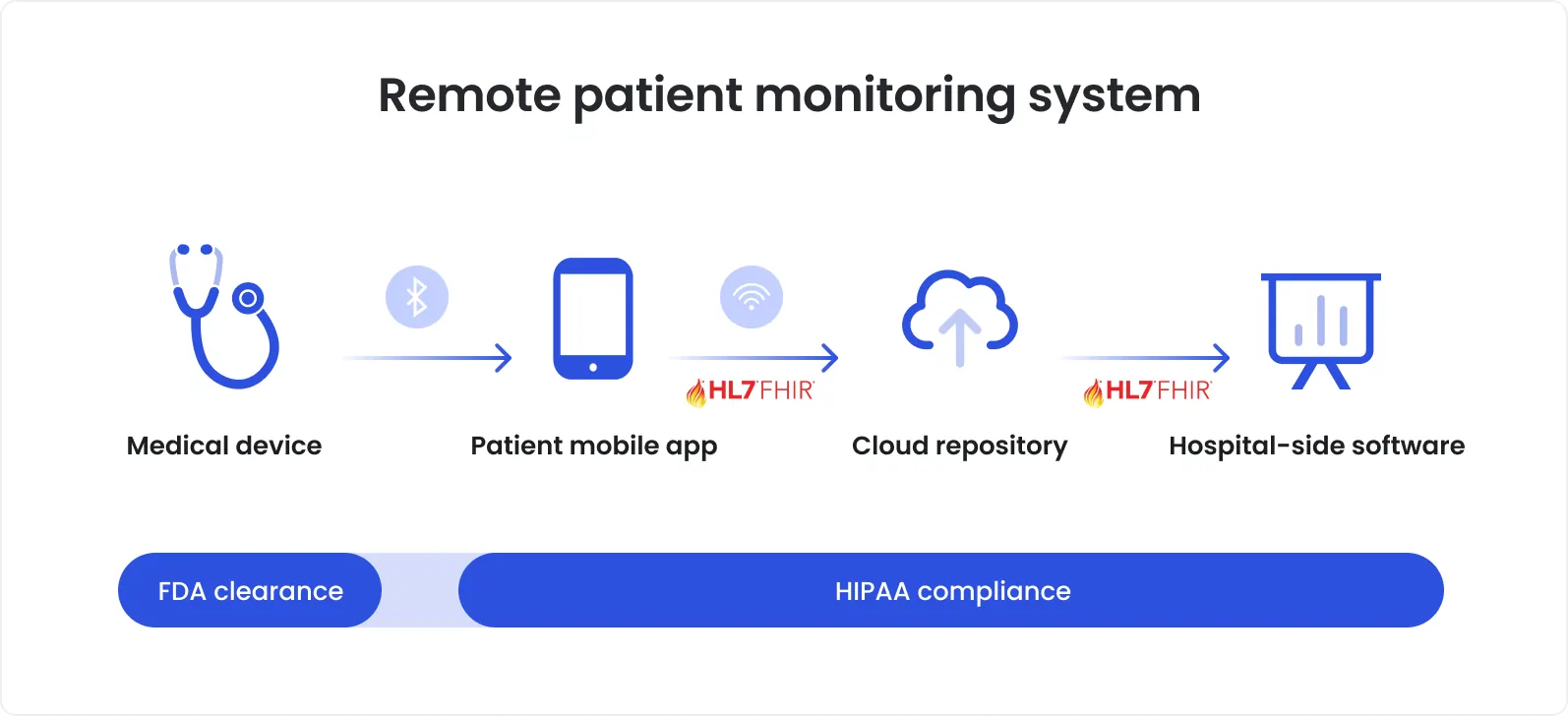
Device risks
Hardware sensors and firmware often store cached data locally. Weak encryption or poorly protected storage can expose protected health information (PHI) if the device is lost or tampered with.
App risks
Mobile apps that sync data often introduce risks because of insecure session handling, weak authentication mechanisms, or unsafe data caching.
Cloud risks
APIs and backend services ingest and process huge data volumes. Misconfigured endpoints, weak authentication, or insufficient audit logging leave room for unauthorized access or silent data manipulation.
EHR integration risks
Data transferred into clinical systems can be tampered with if channels are not properly secured. Improper validation, incorrect code mapping, or poor access controls can result in patient data corruption or disclosure to third parties.
These risks cannot be mitigated with a single session of penetration testing at launch. System security must be carefully assessed throughout development, with QA teams continuously executing security checks.
Tips:
Implement security-by-design validation so that QA engineers review security controls from the earliest design stages.
Prepare threat modeling for each layer.
Introduce SBOM-driven checks to validate all open-source components for known vulnerabilities and update paths.
Verify encryption in motion and at rest across every layer.
Review API configurations to ensure least-privilege access and prevent accidental overexposure.
Confirm that patient data cannot be easily extracted in the case of a device loss or theft.
Set up continuous security regression testing by integrating automated checks for authentication, encryption, and API security into the same pipelines that run functional tests.
Challenge 4: Meeting regulatory standards
RPM systems handle health information and, in many cases, qualify as medical devices. Consequently, they fall under different regulatory frameworks. Depending on the target market, compliance testing may need to ensure the adherence of an RPM solution to:
HIPAA or GDPR for data protection.
FDA 21 CFR Part 820 for medical device quality.
ISO 13485 for quality management.
IEC 62304 for medical device software lifecycle processes.
Notably, having all your tests passed doesn’t guarantee getting approval. Absence of structured documentation can delay certification or even necessitate rework cycles that add months to timelines. That’s why QA is compliance-critical for RPM solutions.
QA teams must create the audit-ready artifacts that are provided to regulators. In these terms, traceability matrices, validation plans, executed test cases, and test logs are just as essential as tests themselves. They serve as evidence that the device was developed under controlled processes.
Preparing compliance artifacts at the end of development is a big mistake because gaps are inevitable, and recreating evidence under time pressure is hardly possible.
Appropriate medical device software testing should inject regulatory requirements into QA activities from the very beginning, ensuring the alignment of every design decision, test, and result with engineering needs and applicable regulatory requirements.
Tips:
Design and maintain a traceability matrix where each regulatory requirement is mapped through design, implementation, and test results.
Create validation plans and verification protocols early to meet functional and safety requirements.
Ensure test results and logs are complete, version-controlled, and auditable.
Implement controlled defect management with every bug linked to the corresponding requirements and retests.
Create risk-based testing strategies that show regulators how potential harms were identified and addressed.
Regularly review and update QA documentation to keep it audit-ready.
Talk to QA experts before risk becomes cost
Challenge 5: Preventing regressions in critical workflows
RPM systems continuously evolve with features multiplying, integrations growing, apps getting updated, and device firmware being patched. However, any code change can cause unforeseen effects.
Because of the high interconnectivity, even a small tweak in a codebase may have significant consequences. A minor adjustment to alert logic could suppress notifications for thousands of users. A slight change in device synchronization may introduce delays that impact the entire pipeline.
With each release, the risk of breaking a critical workflow is high. QA teams must ensure extensive test coverage across all high-impact paths, paying particular attention to data ingestion pipelines, alert thresholds, and device sync processes.
Automated software testing in healthcare is an efficient way to prevent regressions. For example, collaborating with Abbott on a web platform for a sensor-based glucose monitoring solution, the DeviQA team built an extensive automated regression testing suite.

This test suite comprises 1,700 tests, supports 4 web browsers and 20+ devices, and covers all high-risk workflows and critical user journeys, ensuring that updates don’t affect patient safety. Additionally, this approach made regression cycles 18x faster while maintaining 90% test coverage.
Tips:
Create and maintain a map of critical workflows and high-risk paths.
Implement automated regression suites covering these workflows.
Integrate regression tests into CI/CD pipelines for quick feedback on code changes.
Regularly check if all critical paths are covered with tests, especially after major updates.
Challenge 6: Testing UI/UX in accessibility-sensitive RPM solutions
Apart from a device itself, RPM solutions usually include mobile apps or web portals where individuals track readings, acknowledge alerts, or share data with caregivers. These interfaces are part of the safety chain, and weaknesses here can have the same impact as a faulty sensor.
For older people (68% of current RPM users are over age 65) or those with impaired vision, dexterity, or cognitive function, a small design flaw can be a real risk. A too small font may cause a missed alert. Tap targets placed too close to each other can result in taking the wrong action. Poorly structured flows can create confusion when clarity and speed are essential.
Therefore, when it comes to quality assurance for a medical device, thorough UI/UX testing, accessibility testing, and usability testing need to be executed to ensure patient safety and trust.
Tips:
Conduct accessibility audits based on WCAG standards and healthcare-specific usability guidelines.
Run usability tests with elderly and impaired users to spot issues that automated checks cannot detect.
Run cross-device visual checks to test layouts and readability on a wide range of devices, from small budget phones to tablets and desktop portals.
Test interaction flows under stress, for example, one-handed use, slower tap speeds, or with assistive technologies enabled.
Validate support for screen readers and voice commands to ensure critical actions are not blocked.
Regularly review patient workflows, especially after UI redesigns or branding updates.
The right time to bring in QA experts
A common mistake in RPM solution development is involving QA too late. Many teams wait until the MVP is released or key architectural decisions are locked in. At that stage, changes are expensive, timelines slip, and regulatory risk increases exponentially.
Software testing and QA deliver the highest return when embedded early in the development lifecycle. Early QA prevents rework, shortens delivery cycles, and ensures alignment with regulatory and quality standards from day one, rather than retrofitting compliance under pressure.
While QA is essential throughout development, several moments are particularly high-impact:
Architecture planning. Early validation of data flows, security models, encryption, and interoperability assumptions prevents fragile foundations that are costly to correct later.
Pre-FDA or CE submission. Establishing audit-ready documentation, traceability matrices, and validation plans early, and reviewing them before submission, reduces audit friction and approval delays.
Scaling across devices and geographies. Expanding to new hardware, operating systems, or markets multiplies complexity. Structured QA at this stage prevents regressions and preserves system stability.
Post-funding acceleration. After Series A or B, speed becomes a priority. Experienced QA teams enable rapid scaling without introducing uncontrolled quality or compliance risk.
Working with a trusted healthcare QA partner at these points, and continuously throughout development, helps teams avoid rework, reduce release delays, and demonstrate compliance readiness with confidence.
In regulated healthcare software, QA is not a support function. It is a strategic capability that protects investment, accelerates regulatory approval, safeguards product quality, and builds patient trust. Early QA involvement is not a cost, it is risk mitigation and ROI combined.
Why consider DeviQA as your QA partner?
DeviQA is a specialized QA partner for healthcare and medical device software. We support engineering teams at critical moments, early architecture decisions, rapid scaling phases, and FDA or CE readiness.
Our QA approach is built for speed and compliance. We help product teams reduce technical and regulatory risk early, prevent costly rework, and keep delivery predictable as systems grow in complexity and scrutiny.
Get expert input on RPM testing and compliance
Conclusion
To make QA effective in complex RPM environments, teams should focus on a few fundamentals:
design for testability early;
treat compliance artifacts as living assets;
assume scale will break assumptions;
use automation to control risk, not just speed delivery;
align QA ownership with engineering leadership.
DeviQA helps healthcare teams embed these principles into their delivery model, enabling RPM platforms to remain compliant, resilient, and trustworthy as they scale.

About the author
Senior AQA engineer
Ievgen Ievdokymov is a Senior AQA Engineer at DeviQA, focused on building efficient, scalable testing processes for modern software products.